Conditions of the outer or middle ear with conductive or mixed hearing loss
Indications
- Conductive or mixed hearing loss
- Atresia
- Unilateral, bilateral
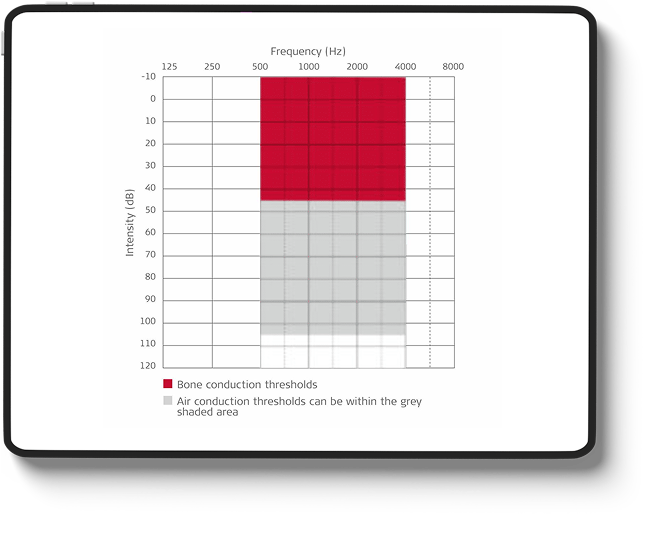
- Stable bone conduction hearing thresholds: ≤45 dB at 500 Hz – see diagram
- Anatomy that allows appropriate placement of the implant
- No retrocochlear or central auditory disorders
- Realistic expectations
- Minimum age: 5 years
Key features of the BONEBRIDGE bone conduction implant system
SAMBA 2 Audio Processor
**The BONEBRIDGE bone conduction implant is MR conditional. Recipients with BCI 602 may be safely scanned at 1.5 Tesla (T) following the conditions detailed in the instructions for use.
**Unless required for diagnostic reasons.
Find a nearby Specialist
Follow us on LinkedIn


